The convergence of four pervasive technologies—ubiquitous sensing, wireless connectivity via smart phones and tablets, cloud computing, and social networking—is rapidly driving the development of “personal” medical electronics for diagnostics, monitoring, and therapeutics. Some call it digital health, e-health, electronically enhanced medicine, m-health (mobile health), or personalized healthcare, emerging as the next generation in healthcare diagnostics and preventive medicine that will be independent of time and place of treatment.
The Role Of Sensors
Personal medical care will depend on a host of sensors embedded in a wearable device or even implanted within the body that will send the patient’s medical information wirelessly to the doctor or healthcare facility. The result will be lower costs, better quality, more convenience, and faster results. Major companies like Apple, Google, and Microsoft are getting into the wearable computing business for wellbeing and healthcare applications.
Related Articles
- Interview: Teresa Sanders Discusses TI Tech That Helps Parkinson's Patients
- Choose The Right Resistors For Medical Applications
- Build A Wrist Heart-Rate Monitor Using An Ultra-Low-Power MCU
Speaking at the Microelectronics Packaging and Test Council meeting in San Jose in May, microelectromechanical systems (MEMS) pioneer Janusz Bryzek, vice president of development for MEMS and sensing solutions at Fairchild Semiconductor Corp., predicted the implementation of e-health in a broad scale of sensing and actuating medical devices such as out-of-body ultrasound and X-ray scanners, dialysis machines, on-the-body wearable devices to monitor health and wellness, in-the-body infusion pumps, and blood glucose sensors.
More than a dozen startups offering wearable devices aimed at fitness and healthcare applications have emerged. Many of these devices are used for personal fitness monitoring, but they also have potential in personalized medical monitoring and diagnostics. For example, the Nike+ FuelBand from Nike Inc. uses wearable sensors and low-power ARM-based cores to track and display what the company calls NikeFuel by counting the wearer’s daily activities (Fig. 1).
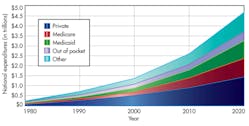
Soaring costs of healthcare expenditures worldwide are driving personal, portable, and wireless healthcare innovations. On a gross domestic product (GDP) capita basis, the U.S. spends about twice the amount of developed countries. According the Office of the Actuary of the U.S. Center for Medicare & Medicaid Services, the U.S. spent about $2.6 trillion in 2010, which is projected to surpass $4.5 trillion by 2020 (Fig. 2).
To empower the development of mobile and wireless healthcare for consumers, Qualcomm formed the $10 million Tricorder XPRIZE competition. The aim is to come up with a tool capable of non-invasively capturing health metrics and diagnosing a set of 15 diseases using the latest advances in wireless sensing, imaging, and portability technologies to replace costlier and bulkier laboratory equipment.
The idea for the Tricorder XPRIZE competition began in late 2010. That’s when Peter H. Diamond, CEO of the XPRIZE Foundation, met with Don Jones, vice president of global strategy and market development at Qualcomm’s Wireless Health Group (now Qualcomm Life Inc.), and Paul E. Jacobs, CEO and chairman of Qualcomm and the Qualcomm Foundation, to kick off the program.
Mobile phone maker Nokia has joined the Qualcomm competition with its own $2.25 million XCHALLENGE, which encourages teams to build sensing technologies for use in identifying and diagnosing diseases. Judges for the competition have chosen 12 innovative hardware and software sensing technologies to advance as finalists in the first challenge, based on technology trustworthiness, privacy and security, standardization, and interoperability.
Wireless and portable medical diagnostics have already been proven in the field. In 2011, the U.S. Food and Drug Administration (FDA) gave its approval to Telcare Inc. to market the first cellular-enabled and portable blood glucose meter, the BGM, which connects diabetes patients with their healthcare providers and families. The BGM works with an FDA-cleared database called Telserve to aid patients in the management of diabetes.
Portable and wireless phones can also be used to monitor patient adherence to prescribed drugs and compliance with dosage directions. “Technology is at the heart of what we do,” claims Aunia Grogan, CEO of Atlantis Healthcare. The company develops and executes interventions to address treatment adherence, which Grogan sees as a rising healthcare cost caused largely by patients’ belief in the effectiveness of their medications and the severity of their illnesses. “The strategic use of technology is critical to effectively deliver mass personalization, ensuring the right person receives the right message at the right time and in the right way.”
One of these technology tools is the Raisin system from Proteus Biomedical. According to the company, patients don’t take 30% to 50% of their prescribed medications, and the costs of hospitalization due to non-adherence are very high. The Raisin system uses a swallowable pill that marries medicine and mobile computing technologies to solve this problem.
Sensors send wireless signals through the body to a receiver that records the type of drug taken, the dose, and the place of manufacture. The system also measures heart rate, body activity, and respiratory rate. Thin-film MEMS layers on each ingestion event maker(IEM) are activated and powered using stomach electrolytes. The system modulates and pulses the current flow to encode information stored in it. It then communicates this data through the body tissue, where a receiver worn on the patient’s skin detects an electric field.
Sensing And Power Challenges
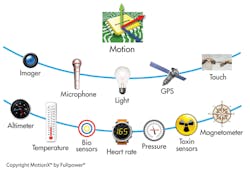
According to Phillipe Kahn, founder of Fullpower Technologies Inc., makers of wearable medical products must overcome three main challenges before their products see greater acceptability in mobile health. They’ll need to be more innovative, create smaller and more accurate sensors, and improve battery performance in terms of smaller size, longer lifetimes, and suitability for wearable medical products. Kahn created the first camera phone in 1997 and is a pioneer in wearable electronics.
Kahn calls his company’s MotionX mobile sensing technology platform, which is embedded in products from Nike, Jawbone, Pioneer, and JVC, the result of innovative thinking from Fullpower Technologies (Fig. 3). “We have a strong focus on breakthrough algorithmic power management that is above and beyond other solutions on the market,” he says. “This applies to wearable computing applications as well as smart phones.”
Kahn also says that many health and fitness wearable mobile medical products can provide readings that are within the 15% to 20% range in accuracy, which paints the wrong picture of a patient’s medical data. He says that levels of 5% accuracy or better are needed. Accuracy can be crucial, for example, in diagnosing lower back pain and gout during a long gait.
BodyMedia claims to have the highest accuracy for a wearable fitness monitor in its FIT calorie counter, with accuracies greater than 90% (Fig. 4). Its on-body multi-sensor platform meets FDA Class II category regulations and conforms to the ISO 13485 standard. More than 150 clinical studies verified this accuracy claim, claims BodyMedia.
Unlike other wearable body monitoring devices, the FIT includes four sensors that provide the right size, cost, and functional relevance working together to predict the most accurate calorie burn information outside of a laboratory. One sensor measures galvanic skin response due to sweating. A three-axis accelerometer measures motion and the number of steps taken. A temperature sensor measures body heat. And, a heat flux sensor measures the rate heat is dissipated from the body.
Samsung Electronics has embarked on a forward-looking vision that is bound to dramatically change wireless mobile health and define the next decade of medical technology. Speaking at this year’s MobileBeat Conference, Samsung’s president and chief strategy officer Young Sohn showed a video demonstrating a foldable form factor for large displays, photonic crystal reflective lighting that requires no backlighting, and RF wave sensors integrated within worn patches that check and transmit a patient’s health data.
Sensors don’t need to be integrated into a wearable device, Samsung demonstrated. Instead, they simply can be used as connected add-on accessories. The folded display form factor allows the expansion of the display screen without drawing additional battery power.
Wearable e-health portable devices require a certain degree of sensor fusion that intelligently combines and processes data streams from multiple sensors, producing an output whose sum is greater than the individual inputs combined. Many sensor fusion products are available in hardware and software packages that are often proprietary to a specific vendor, usually tied to that vendor’s sensor offerings.
Researchers now are looking at data fusion rather than just sensor fusion. Many experts consider this approach the key to enabling more intelligent applications, which will be needed in e-health devices.
Honing Communications Chips
Semiconductor IC manufacturers have long been serving the medical community with highly integrated, low-power, and accurate devices, attributes required in portable and wearable wireless communications products. Renesas Electronics is collaborating with Belgium’s IMEC to advance wireless communications with the first multi-standard RF receiver and an analog-to-digital converter (ADC) CMOS IC manufactured on a 28-nm process. The devices target LTE-Advanced and next-generation Wi-Fi communications.
“Using a 28-nm CMOS process allows us to offer high levels of integration and low power levels, increasingly important parameters for the latest and future wireless communications devices,” explains Renesas’ Nelson Quintana, director of marketing. “We’ve been supplying extremely low-power and highly accurate devices to the medical community such as our RX100 32-bit entry level microcontroller (MCU).”
Silicon Laboratories is acquiring Norway’s Micro Micro AS, one of the first companies to develop 32-bit MCUs based on a licensed ARM-Cortex-M3 core with an emphasis on low-power and battery operated applications. Silicon Laboratories is trying to develop a critical mass in its pursuit of the wireless and embedded Internet of Things (IoT) market, which includes portable medical electronics.
Bluetooth low energy (BLE) is serving as a good connectivity model for e-health devices. It suits applications that send limited data and need to operate from very low power levels, like blood pressure monitors and pulse oximeters. Such e-health devices are more likely to operate at home or in a doctor’s office. But hospital and clinical settings involve larger volumes of data, and medical devices generally operate from larger batteries or an ac power source. That’s also the case for implantable devices like infusion pumps for delivering insulin to the pancreas. Here, Bluetooth is the correct communications choice.
It should be noted that the Bluetooth Smart communications protocol supports BLE but not the classic Bluetooth. The Bluetooth Smart Ready protocol is available to communicate with both BLE and classic Bluetooth, and it is already in use in many smart phones and tablets.
“Our research has shown and all the information is indicating that BLE is the predominant one in the market now. Using BLE components adds a new level of functionality that enhances their power-saving capability,” says Chuck Parker, executive director of the Continua Alliance, a non-profit, open industry organization of more than 200 healthcare and technology companies worldwide collaborating to improve the quality of personal healthcare.
Parker also sees ZigBee as another communications protocol that offers us some capabilities in meshing networks. “It allows us to follow an individual through a home or through a relatively large-square-foot setting. Bluetooth simply can’t accomplish that. It’s got a 33-foot limit, whereas with ZigBee, with its meshing points, we can basically monitor the entire facility, whether it’s on a single floor or multiple floors,” he says.
Medical implants are omnipresent, and many are linked to home networks. According to Janus Bryzek, 600,000 of the 2.5 million medical implants like pacemakers and the like are alreadylinked via home networks for remote sensing and monitoring.
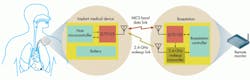
Medical implants such as insulin infusion pumps, cardiac pacemakers and defibrillators, drug pumps, and neuro-stimulators all can use Microsemi’s ultra-low-power medical implantable communication service (MICS). It connects the ZL70102 wireless RF transceiver chip in a module implanted in a patient’s body to a module within an external monitoring basestation (Fig. 5). MICS operates in the 402-MHz to 405-MHz band and consumes less than 6 mA in the transmit/receive modes, 290 nA in the listen before transmitting mode, and just 10 nA in the sleep mode. Micosemi’s ZL70321 implantable implantable radio module works with the ZL70102 and has an integrated matching network, a surface acoustic-wave (SAW) filter for unwanted blockers, and an extra low-noise amplifier for maximized receiver sensitivity.
Researchers at Switzerland’s Centre Suisse d’Electronique et de Microtechnique (CSEM) are working on the WISERBAN project for much smaller implantable medical devices that will feature smarter communications and consume less power. This project will develop an ultra-miniature wireless body area network (BAN) comprising a 2.4-GHz radio, a microprocessor for sensor data processing, and RF MEMS devices for improved radio performance, all within a 4- by 4- by 1-mm system-in-package (SiP) housing, and consuming just a few milliwatts. According to Vincent Peiris, section head of CSEM’s analog and IC design section and the project’s coordinator, the group is developing devices that are 50 times smaller and require 20 times less power than existing consumer products.