Innovative Breathalyzer Calibration Instrument Blends Optics, Electronics
It’s a question with which many engineers wrestle: “Is my reading as accurate as it reads?” It’s especially challenging for instrumentation that must assess a real-world physical variable using an external sensor. Do you qualify your own unit by checking it against a “better” standard instrument and arrangement? How do you know if that one really is better? Is it based on a more carefully calibrated version of your instrument, or does it use a radically different principle and thus provide an independent check on the results?
This is the problem faced by users of the breathalyzer, a generic term for the portable, non-invasive instrument that determines a person’s “blood-alcohol” level (specifically, ethyl alcohol or ethanol) in real time based on a breath sample. These units are widely used in the field by law-enforcement officials, and their calibration and accuracy are often challenged in court.
There are two very different ways to calibrate these units—one using a certified dry ethanol-air mixture, the other using generation of an ethanol-air-water vapor mixture at a known temperature. Each has major practical drawbacks in convenience and consistency.
To address this issue, a Swiss-based team at Empa, a 125-year-old materials-testing institute associated with ETH Zurich (formerly called the Zurich Federal Polytechnical School), has developed a calibration system based entirely on an electro-optical approach. The details of the underlying optical principles and unit’s construction are discussed in their Optics Express paper “High-precision ethanol measurement by mid-IR laser absorption spectroscopy for metrological applications.” The electronics are covered in further detail in a second paper, “Laser driving and data processing concept for mobile trace gas sensing: Design and implementation,” in Review of Scientific Instruments.
The unit uses a quantum cascade laser (QCL) as a mid-band IR source to take advantage of the strong fundamental absorption features of the molecules of interest (Fig. 1). Some available commercial ethanol analyzers use non-dispersive infrared (NDIR) technology, but the coherent radiation of a laser source supports an extended-length optical path of tens of meters via a multipass cell. This long path increases the absorption signal and sensitivity, while also improving SNR due to the much brighter IR radiation of the laser sources compared to conventional IR light sources. Further, the narrow bandwidth of the laser emissions can be rapidly scanned across the optical band, resulting in highly selective and sensitive measurements of the absorption.
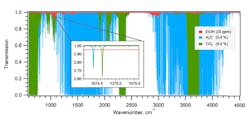
1. Shown is simulated mid-IR transmission spectra of 25 ppm ethanol, 5.4% water, and 5.0% CO2 at gas pressure of 100 hectoPascals (1.45 psi), room temperature, and absorption path of 25 meters. The inset reveals the spectral window selected for precise measurement of ethanol. The ethanol data is from the U.S. Pacific Northwest National Laboratory (PNNL) IR database (http://nwir.pnl.gov) and adjusted according to the experimental conditions. (Source: Empa, Laboratory for Air Pollution and Environmental Technology)
The entire unit consists of two parts: an “upper level” with the laser, multipass cell, optical detector, and related optical elements; the electronics unit is immediately below it (Fig. 2). The latter includes the power supply, an embedded computer, a temperature controller for the optical module temperature stabilization via a Peltier unit (the laser and the optics module heat sinks are maintained a 19.00 ± 0.05°C), extremely low-noise laser-driver electronics, and an FPGA-based system-on-chip hardware-control and data-acquisition module with a sampling rate of 125 Msamples/s and 14-bit resolution.
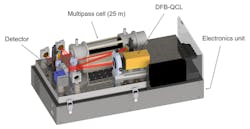
2. Here’s a 3D CAD drawing of the QCLAS for the ethanol measurement (without the cover); the red line represents the path of the IR laser beam. The enclosure beneath the optical breadboard contains the entire electronics hardware. The dimensions of the instrument with the cover are 33 × 30 × 63 cm3. (Source: Empa, Laboratory for Air Pollution and Environmental Technology)
Any excess noise from the laser-injection current leads to temperature changes (∆T) in the active region of the dual QCLs (a pair of QCLs are used for path balance) and consequent drift in its emission frequency (∆ν). For the laser-driver control and basic data processing (LabView provided additional processing), they used an inexpensive FPGA-based platform from Red Pitaya to implement a digital lock-in amplifier. That platform is based on a Xilinx ZYNQ-7010 processor and includes an integrated dual-core Arm Cortex-A9 processor with Xilinx 7-series FPGA logic.
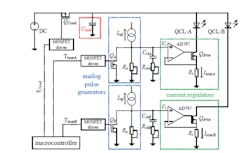
3. This is a schematic of the dual-channel intermittent continuous-wave (ICW) QCL current driver. An external dc power supply charges Cload (red). Shortly before a laser is turned on, Cload is disconnected from the external power supply and serves as a power source for the corresponding laser. The laser current (up to 1 A) is provided by the current regulators (green), while the amplitude and shape are defined by the settings of the analog pulse generator (blue). The temperature coefficient for all of the resistors and capacitors are ±5 ppm/°C and ±25 ppm/°C, respectively. The microcontroller triggers the different phases according to the timing diagram defined by the user. (Source: Empa, Laboratory for Air Pollution and Environmental Technology)
The current-source laser-driver design (Fig. 3) delivers 1 A for about a 200-μs time duration in what’s called ICW (intermittent continuous wave) driving It has three key features:
• The driving current for the QCLs is provided by an op-amp-based current regulator, which enables active and independent current control
• A high-precision analog pulse generator is designed to generate current pulses with adjustable amplitude, shape, and width
• Similar to a previous design, the electrical power of the driving circuit is provided by a load capacitor, which is disconnected from the external power supply during the operation of the QCLs for the purpose of noise reduction. Since the drive current is generated by the current regulator, it’s independent of the voltage level on the load capacitor and allows use of a relatively small capacitor (330 μF), reducing noise and improves thermal stability.
Assessed performance of the unit is defined by factors such as accuracy, precision, stability, linearity, and cross-sensitivities to changes in various parameters, among others. The team characterized these using precision gas sources, gas-flow controls, and gas-mixing units from laboratory-certified suppliers, all traceable to higher standards, to develop the various concentrations with both static and dynamic attributes.
For example, they evaluated the instrument response to changing ethanol concentration within the range of 25 to 225 ppm, in steps of 25 ppm by diluting 225 ppm ethanol with synthetic air. The highest concentration in the range corresponds to about 0.8% blood alcohol content (BAC). The linear-regression model shows a very tight correlation (Fig. 4).
4. Measured ethanol concentration as a function of sampled ethanol concentration in the range of interest shows a high degree of linearity and correlation. (Source: Empa, Laboratory for Air Pollution and Environmental Technology)
The two papers cited above provide much more detail on the gas-related chemistry; selection of the optimum optical window; compensations and corrections for gas concentration, temperature, and other factors; and details of the interaction between laser and optical issues due to concentration levels and absorption. In addition, many details of the electronic circuitry—such as enhancement to the laser-drive implementation to ensure highest stability and lowest noise—are also discussed in their published papers.