Maxim’s Wearable Health Sensor Platform Adds a New Dimension
Maxim Integrated Products’ first Health Sensor Platform (HSP) made it much easier to build wearable devices. However, the latest version—HSP 2.0 (Fig. 1)—lets developers monitor electrocardiogram (ECG), heart rate, and body temperature. The modular system is designed to handle upgrades or enhancements, which in the long run can save developers months of design and implementation work.
1. Maxim’s Health Sensor Platform 2.0 (left) can monitor ECG, heart rate, and skin temperature. The device can be taken apart (right) to enable changes or upgrades to the sensor system.
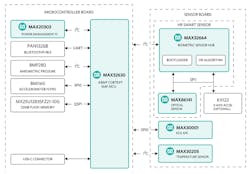
The $399 HSP 2.0 contains a number of Maxim Integrated’s components, starting with the Darwin low-power MAX32630 ARM Cortex-M4F microcontrollers. The platform is tied into a MAX32664 sensor hub (Fig. 2) that handles a range of sensors, including the MAX86141—an optical sensor that tracks heart rate as well as being a pulse oximeter. The MAX30001 analog front end (AFE) handles the ECG sensors, while the MAX30205 handles temperature to within 0.1°C. Maxim also covers power management with its MAX20303 PMIC, which is critical for such mobile devices.
2. The HSP 2.0 is built around Maxim’s MAX32630 ARM Cortex-M4F tied to a MAX32664 sensor hub that handles a range of sensors.
In addition, the HSP 2.0 uses third-party chips and modules, including the Bluetooth support. The system is charged and managed with a USB Type-C connection. The compact module has an expansion header and display interface and contains the processor and Bluetooth support (Fig. 3).
ARM Mbed supports the MAX32630 microcontroller. This provides access to a large, open-source library of tools as well as a development and IoT communication framework. System software support for over-the-air updates is in the works. The MAX32630 also includes a secure element that can provide encryption and secure key support. An Android application is also provided that allows for remote viewing of data.
3. The processor module is compact with an expansion header.
The platform doesn’t have FDA approval as a medical device, but a developer could use it as the basis for such a product. It’s a matter of getting the appropriate approvals so that data could be utilized by medical professionals. Of course, the device can be used to acquire and provide information to users.
Maxim Integrated has put together an amazing package that opens opportunities for professional developers and makers. There’s no comparable platform available elsewhere. Its modular approach enables future hardware upgrades as well as custom sensor arrays without incurring major redesign costs.
About the Author
William G. Wong
Senior Content Director - Electronic Design and Microwaves & RF
I am Editor of Electronic Design focusing on embedded, software, and systems. As Senior Content Director, I also manage Microwaves & RF and I work with a great team of editors to provide engineers, programmers, developers and technical managers with interesting and useful articles and videos on a regular basis. Check out our free newsletters to see the latest content.
You can send press releases for new products for possible coverage on the website. I am also interested in receiving contributed articles for publishing on our website. Use our template and send to me along with a signed release form.
Check out my blog, AltEmbedded on Electronic Design, as well as his latest articles on this site that are listed below.
You can visit my social media via these links:
- AltEmbedded on Electronic Design
- Bill Wong on Facebook
- @AltEmbedded on Twitter
- Bill Wong on LinkedIn
I earned a Bachelor of Electrical Engineering at the Georgia Institute of Technology and a Masters in Computer Science from Rutgers University. I still do a bit of programming using everything from C and C++ to Rust and Ada/SPARK. I do a bit of PHP programming for Drupal websites. I have posted a few Drupal modules.
I still get a hand on software and electronic hardware. Some of this can be found on our Kit Close-Up video series. You can also see me on many of our TechXchange Talk videos. I am interested in a range of projects from robotics to artificial intelligence.
Voice Your Opinion!
To join the conversation, and become an exclusive member of Electronic Design, create an account today!

Leaders relevant to this article: