Flexible instrumentation supports medical device test
Designers of battery-powered medical devices require low-noise power sources as well as precision measurement capability, according to Bob Green, senior market development manager at Keithley Instruments. Wireless medical devices, he said, include deep-brain neurotransmitters, cochlear implants, gastric stimulators, cardiac defibrillators and pacemakers, foot-drop implants, and insulin pumps.
A Keithley white paper points out that portable medical devices, whether worn on the body or implanted, can alleviate pain and treat conditions including cardiac arrhythmia, hearing and vision impairment, movement disorders including Parkinson’s disease and epilepsy, and even obsessive-compulsive disorder. Such devices often are powered by inductive RF links, and even ones with batteries may need to be periodically recharged.
Green traced the emergence of requirements for low-power designs for such devices. In the 1950s, he said, pacemakers, for example, were worn outside the body, and power consumption wasn’t a key issue. But the devices have evolved into implantable pacemakers.
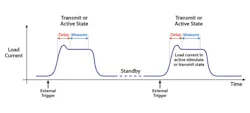
He noted that such a device can have drastically different power requirements depending on the mode in which it is operating—for example, standby mode, stimulate mode, or transmit mode (in which it wirelessly transmits status information from within the body). An implantable defibrillator, he said, might draw 0.5 µA in sleep mode but 5 A in active mode.
In all cases, designers must answer two questions, Green said. First, is the standby current low enough to meet power life specifications? And second, is the stimulation current within specification to minimize power consumption?
The combination of modes requires instrumentation that can make both low-current and pulse measurements, Green said. The white paper elaborates, noting that a low-current measurement circuit requires a sensitivity of 1 µA or even 0.1 µA. Low-current measurements can employ filtering and can occur over many power-line cycles.
Pulse measurements take place over a very short period of time—perhaps a few hundred microseconds—and must be synchronized with the device under test or its control system. In addition, it’s important to measure the right portion of the pulse, which can require insertion of a delay to avoid measuring overshoot. In addition, the instrumentation must provide fast response to detect voltage drops that drastic load changes can cause.
The differing measurement requirements for active and sleep modes might suggest the need for multiple instruments, including perhaps a DMM, a source-measure unit, and an oscilloscope. However, Keithley is aiming to help meet the challenges posed by medical device design with its recently introduced Series 2280S precision measurement, low-noise, programmable DC power supplies.
Green said that unlike conventional power supplies, the Series 2280S power supplies also are sensitive measurement instruments with the speed and dynamic range essential for measuring standby current loads and load current pulses. They can supply up to 192 W of low-noise, linear-regulated DC power; the Model 2280S-32-6 can generate up to 32 V at up to 6 A while the Model 2280S-60-3 can provide up to 60 V at up to 3.2 A.
Unlike conventional power supplies, Series 2280S supplies can make voltage and current read-back measurements with up to 6½ digits of resolution for maximum precision or as low as 3½ digits for greater speed, allowing engineers to optimize speed and accuracy for their applications. Load currents from 100 nA to 6 A can be monitored with high accuracy, enabling the instrument to test a device in active as well as standby modes. Four load-current measurement ranges (10 A, 1 A, 100 mA, and 10 mA) support measuring full-load currents, standby-mode currents, and small sleep-mode changes precisely.
For monitoring fast-changing and pulse-like load currents, Series 2280S supplies can capture dynamic-load currents as short as 140 μs to monitor load currents easily in all operating modes for determining total power consumption of the device.
The supplies also include a graphing function that simplifies monitoring the stability of the load current, capturing and displaying a dynamic load current, or viewing a start-up or turn-off load current. These power supplies can store up to 2,500 measurement points and compute statistics on the stored data. Statistical calculation options include average, maximum, minimum, peak-peak, and standard deviation. The instruments feature a list mode function; up to nine lists of sequenced voltage levels can be created and saved with up to 99 distinct voltages in each list. A single trigger will automatically execute the list once or multiple times. To minimize test times in automated systems, an external trigger input allows hardware synchronization and control by other system instruments.
Of course, the new instrument is applicable to products in addition to wearable and implantable medical devices. Green outlined how products have evolved with battery life being a key spec. He pointed out that a Galaxy 1 phone offered 75 hours of standby power. The S4, in contrast, provides 370 hours. Operating modes have become more efficient as well. He noted that an Android 4.4 offered 345 minutes of Wi-Fi browsing; the Android L offers 471 minutes.
And like pacemakers, cellphones have low-power sleep modes but may draw considerable current when active. An LTE phone may draw 38 mA in sleep mode but 580 mA when active, Green said, making the 2280S useful in their design and test.
Reference
Tomorrow’s Wireless, Portable, and Implantable Medical Devices Require High Precision Characterization, Keithley Instruments, White Paper, 2014.
For more information
Keithley Instruments
About the Author
Voice Your Opinion!
To join the conversation, and become an exclusive member of Electronic Design, create an account today!

Leaders relevant to this article: