High levels of low-density lipoprotein, or LDL cholesterol (LDL-C), have long been associated with increased risk of cardiovascular (CV) disease. In fact, as explained in the introductory remarks accompanying a recent FDA briefing document, “… For more than two decades, the FDA has used a reduction in LDL-C as a surrogate for CV risk reduction for several lipid-altering drugs to support traditional approval. Certainly, at least for statins, the validity of a reduction in LDL-C as a surrogate for reduced CV risk has been confirmed through numerous randomized controlled trials involving multiple drugs in the class and a variety of patient populations with varying degrees of baseline risk and LDL-C values.”
The report continued, “The FDA has defined a surrogate endpoint as a marker such as a laboratory measurement or physical sign that is used in therapeutic trials as a substitute for a clinically meaningful endpoint that is a direct measure of how a patient feels, functions, or survives and is expected to predict the effect of therapy.”1
In some cases, manufacturers conducted additional studies to determine the reduction in CV risk associated with the drug in question. However, as reference 1 stated, “With such a traditional approval [a statin approval based only on its LDL-C level reduction capabilities], FDA does not have a regulatory mechanism to require any further efficacy trials.”
A recent study analyzed the use of statins in the United States. The authors found, “Of the predictors of statin use, hyperlipidemia [elevated lipid level] had a much greater association with statin use than the other predictors. Peripheral arterial disease and tobacco use were not significant. In the primary prevention cohort [persons in the 16,712 study population without a diagnosis of diabetes or heart disease], individuals with a diagnosis of hyperlipidemia had a higher probability of being on a statin, 30%, than individuals without a diagnosis of hyperlipidemia, 0.6%.”
The paper concluded, “… Hyperlipidemia appears to be the driving force behind statin use…. For years, guidelines, general public health messages, and direct-to-consumer advertising have anchored statins to cholesterol reduction rather than CV risk reduction.”2 Unfortunately, about 5.6 million individuals in the United States with coronary artery disease and about 9 million with diabetes and older than 40 years are not regularly using statins for a variety of reasons.
Statin backlash
Statins generally reduce the risk of CV disease in patients already diagnosed as having a significant risk. However, not everyone can tolerate a statin. Myalgia, fatigue, transaminitis (elevated levels of certain liver enzymes), and memory problems are among the adverse effects. Up to about 10% of the population analyzed in reference 2 could not or would not take a statin. According to a 2014 article, “… In the next 10 years, we’re going to see a backlash against statins. In fact, we’re starting to see it already. We’re seeing now that they may not benefit everybody we thought they would. And, we’re seeing that there are some serious risks involved.”3
In the U.K., the National Institute for Health and Care Excellence (NICE) has recommended that about half of the adult population aged 50 and older should be taking statins. A number of studies have been cited as supporting this recommendation. Results of another study that linked Parkinson’s disease and 46% increased risk of diabetes to statin use were highlighted in a recent Express newspaper article.
The article explained that the Parkinson’s study was conducted over a period of 20 years and involved nearly 16,000 patients. It found that “… those who take the cholesterol-lowering drugs are more than twice as likely to develop Parkinson’s disease later in life than those who do not.” According to Dr. Kailash Chand, deputy chairman of the British Medical Association, but speaking on his own behalf, “The risks of side effects of these drugs are far greater than any potential benefits [for low-risk patients], and it is high time these drugs were restricted in the low-risk population.”
The findings are significant because the NICE recommendations include a 10-year plan with the goal of prescribing statins even for low-risk patients with “… just a 10% chance of a heart attack within a decade.” The article continued, “The findings have alarmed experts who say if applied to the number of Britons deemed eligible for statins, it could equate to 150,000 extra patients with Parkinson’s, a central nervous system disorder affecting one in 350 mostly older people.”4
Changing the guidelines
In recognition of what may have been misplaced emphasis on lower LDL-C values, the American College of Cardiology (ACC) and the American Heart Association (AHA) jointly released new cholesterol guidelines in 2013. The intention was to move the focus toward reducing the risk of CV disease and away from lowering LDL-C values to some target number. Unfortunately, because of the method used to assess CV disease risk and the associated limits, “The new ACC-AHA guidelines substantially broadened the number of individuals for whom statins are recommended, primarily by broadening the eligible population to lower levels of cardiac risk.”2
The authors commented that, “Most newly eligible individuals in the guidelines are those who do not have a diagnosis of coronary artery disease. Benefits from statins … in terms of a reduction in adverse CV events and in mortality are controversial and at best small.”2 Basically, statins are being recommended for people who probably do not need them while at the same time millions of people with diagnosed CV disease are not taking statins.
Making further tools available
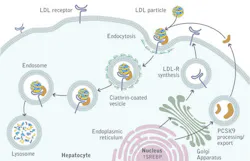
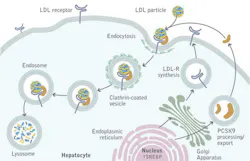
Statins work by partially inhibiting cholesterol production. A new class of monoclonal antibody drugs called PCSK9 inhibitors reduces the PCSK9 protein in the liver, allowing the liver to remove more of the LDL-C circulating in the bloodstream. As shown in the figure and described in reference 1, “A complex of LDL-C, LDLR [LDL receptors] and PCSK9 is internalized into hepatocytes into clathrin-coated pits and subsequently undergoes lysosomal degradation.”
When a PCSK9 inhibitor is used in conjunction with statins, the LDL-C reductions can be very large—40% to 60%. This means that instead of patients striving to reduce their LDL-C level to a little less than the average 100 mg/dl in healthy people, it’s now possible to achieve levels of 40 or 50 mg/dl. Does such a low level make sense? Is there really a correlation between extremely low LDL-C values and greatly reduced risk of CV disease?
Two PCSK9 inhibitors were approved in 2015 by the FDA: Amgen’s Repatha (evolocumab) and Praluent (alirocumab) from Sanofi US and Regeneron Pharmaceuticals. Both drugs are injected, once every two weeks or once a month for Repatha, or once every two weeks for Praluent. An article that reviewed some of the concerns expressed about Repatha by members of the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee (EMDAC) helps explain the approvals process.
Eleven of the 14 members thought that the benefits of lowering LDL-C outweighed the risks for patients with HeFH—a gene mutation that affects the LDL-C receptors in the liver. In contrast, all 14 members recommended use for patients with HoFH, a much rarer and less well-controlled condition. A large 27,500-patient study to determine the efficacy and safety of Repatha is underway and expected to be completed in 2017. So, one concern for the EMDAC members was whether to approve the drug now at least for limited use or to wait until the study was complete.
As explained in the article, some of the members who were uncertain about approving the drug “… might have been more reassured if evolocumab was reviewed as part of the FDA’s accelerated approval program.”
According to a Medscape article, “Instituted in 1992, the accelerated approval program was designed to approve drugs to treat serious conditions on the basis of a surrogate end point. As part of this pathway, pharmaceutical companies are required to complete phase 4 confirmatory studies, and if such trials turn out negative, the FDA has regulations in place to remove the drug from the market. In contrast, Amgen would not be required to show a positive effect on cardiovascular outcomes if evolocumab were approved on the basis of changes in LDL cholesterol.”5
Dr. William Hiatt, University of Colorado School of Medicine, Aurora, commented, “Without the [accelerated approval] option, a yes vote means that we’re going to expose a large population of patients to a drug that has a very limited safety base and absolutely no clinical evidence of benefit.” And, Dr. Peter Wilson, Emory University, Atlanta, GA, also voted against general approval, noting the lack of long-term data. He said, “’I’m particularly concerned if the agency were to approve the drug, there’d be a large number of people [who receive the drug] in whom there’d be very little benefit.’”5
Part of the EMDAC discussion focused on the appropriateness of lowered LDL-C as a surrogate for better CV health. In general, most members thought it made sense because of the good results statins have produced for many people. On the other hand, one member remembered that niacin had lowered LDL-C but had failed in two large CV-outcome studies.
As stated in an FDA announcement, “Praluent is approved for use in addition to diet and maximally tolerated statin therapy in adult patients with HeFH or patients with clinical atherosclerotic cardiovascular disease such as heart attacks or strokes, who require additional lowering of LDL cholesterol.”6
“Repatha, the second drug approved in a new class of drugs known as PCSK9 inhibitors, is approved for use in addition to diet and maximally tolerated statin therapy in adult patients with HeFH, HoFH, or clinical atherosclerotic cardiovascular disease, such as heart attacks or strokes, who require additional lowering of LDL cholesterol.”7
Conclusion
Because the FDA continues to support the use of statins based both on their capability to lower LDL-C levels and the corresponding reduction in CV disease risk, reference 1 concluded, “… One clinical scenario that may seem practical for monotherapy is the use in patients who cannot tolerate statins.” In other words, PCSK9 inhibitors could be used as an alternative treatment for statin-intolerant patients.
However, reference 1 then digressed for a few paragraphs that questioned the number of patients who actually could not take statins. “The division’s working definition of statin intolerance for the purpose of exploring this concept in clinical trials of novel non-statin LDL-C lowering therapies follows: The inability to tolerate at least 2 statins: one statin at the lowest starting daily dose, defined as rosuvastatin 5 mg, atorvastatin 10 mg, simvastatin 10 mg, lovastatin 20 mg, pravastatin 40 mg, fluvastatin 40 mg or pitavastatin 2 mg, AND another statin at any dose, due to the prespecified intolerance symptoms (such as skeletal muscle, hepatic, psychiatric, or cognitive-related symptoms) that began or increased during statin therapy and stopped when statin therapy was discontinued.”
The use of lowered LDL-C as a surrogate is both clearly stated and also questioned in reference 1 as well as in reference 4, which included the reasoning of EMDAC dissenting members. In addition, the joint ACC/AHA guidelines, although perhaps too inclusive, attempt to shift the measure of drug efficacy from lowered LDL-C to reduced CV disease risk.
Repatha and Praluent have only just started to be used, and long-term safety and efficacy studies have years to run. They have been restricted to only HeFH patients for Praluent or HeFH and HoFH patients for Repatha. No doubt, as the results of further PCSK9 and statin studies become known, the risk/benefit balance associated with cholesterol-lowing drugs will continue to be debated.
References
- “FDA Briefing Document,” Endocrinologic and Metabolic Drugs Advisory Committee (EMDAC), June 2015.
- Johansen, M., E., et al, “Cardiovascular Risk and Statin Use in the United States,” The Annals of Family Medicine, May/June 2014, Vol.12, No. 3, pp. 215-223.
- Hyman, M. MD, “Should I Stop My Statins?,” The Huffington Post, Jan. 7, 2014.
- Johnston, L., “EXCLUSIVE: Parkinson’s link to statins: Calls to end widespread use of the drug,” Express Newspapers, April 27, 2015.
- O’Riordan, M., “Approve PCSK9 Inhibitor Evolocumab, FDA Panel Recommends,” Medscape, June 10, 2015.
- “FDA approves Praluent to treat certain patients with high cholesterol,” FDA news release, July 24, 2015.
- “FDA approves Repatha to treat certain patients with high cholesterol,” FDA news release, Aug. 27, 2015.
About the Author

Tom Lecklider
Voice Your Opinion!
To join the conversation, and become an exclusive member of Electronic Design, create an account today!

Leaders relevant to this article: